This web page was produced as an assignment for Genetics 677, an undergraduate course at UW‐Madison.
3D structures of the BLM protein domains
I was unable to find any predicted 3D structure of the whole BLM protein in any of the proteomic databases. Thus, I started to search on primary literature database like PubMed to look for recently solved BLM protein structure. Instead of obtaining structure of the entire BLM protein including every domains, I found only structures of individual domain- HRDC and RecQ helicase. According to Sato et al., structures of the BLM protein are not yet well studied due to difficulties in protein purification and crystallization. Therefore, only few domains such as the HRDC domain were successfully solved via NMR spectroscopy despite extensive structural studies have been conducted [1]. Below is a compilation of the predicted structures of different domains.
Paper: Solution structure of the HRDC domain of human Bloom syndrome protein BLM [1]
Akiko Sato, Masaki Mishima, Aki Nagai, Sun-Yong Kim, Yutaka Ito, Toshio Hakoshima, Jun-Goo Jee and Ken Kitano
|
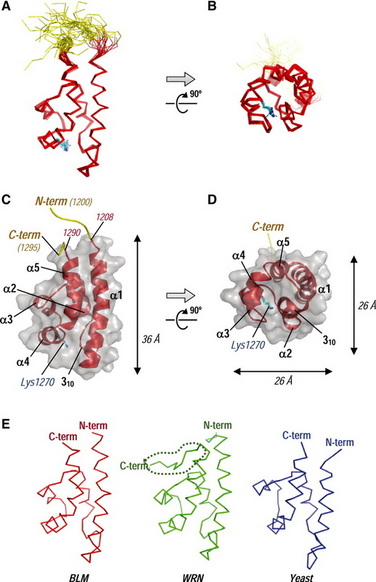
Figure 1. This is a recently published structure of the HRDC domain of human BLM protein by Sato et al.. Panel A shows the "front view of the backbone superpositions of the final 20 simulated annealing structures of the BLM HRDC domain" . The HRDC domain core is represented in red color, while the distorted N- and C-terminal residues are in yellow [1].
Panel C shows the front view of BLM HRDC as a ribbon model. According to Sato et al., this model of structure has the lowest energy among all the structures determined by NMR technique. The height of BLM HRDC is approximately 36 Å, while the width and depth are 26 Å [1]. The authors also attempted to compare the structures of BLM HRDC with WRN HRDC and Saccharomyces cerevisiae Sgs1 (panel E). In fact, BLM and WRN are members of the RecQ helicase superfamily. BLM and Sgs1 HRDC lack the C-terminal structured loop that is present in WRN HRDC (encircled by dashed line) [1]. |
|
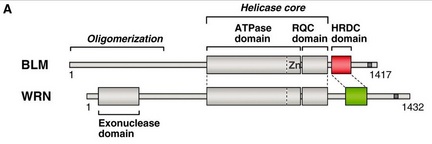
Figure 2. Another diagram from the same paper shows the location of the HRDC domain and helicase core in the BLM protein. The helicase core is comprised of the ATPase domain and RQC domain. The HRDC domains is coloured in red (BLM) and green (WRN). The nuclear localization signal is depicted as a shaded bar near the C-terminus of the BLM protein. Notably, the entire sequence identity between BLM and WRN is approximately 15%, while the identities within the ATPase, RQC and HRDC domains are approximately 30, 10 and 20%, respectively [1].
|
Paper: Structural and functional analyses of disease-causing missense mutations in Bloom syndrome protein
Rong-Bing Guo, Pascal Rigolet, Hua Ren, Bo Zhang, Xing-Dong Zhang, Shuo-Xing Dou, Peng-Ye Wang, Mounira Amor-Gueret, and Xu Guang Xi
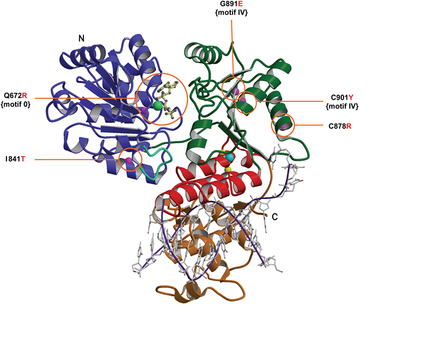
Figure 3. The BLM helicase catalytic core is represented in a ribbon drawing while five mutation sites are mapped on the modelled BLM helicase structure. The authors compared the BLM protein amino acid sequence to that of E. coli RecQ helicase and modelled the structure of BLM helicase core based on the structure template of E. coli RecQ helicase [2].
Analysis
It was clear that the structure of BLM protein is yet to be solved. Researchers have tried different approaches such as building individual protein domain based on structure template of members of RecQ helicase superfamily and utilizing NMR spectroscopy to predict the BLM domian 3D structures. I think it is essential to solve and characterize the BLM protein structure because it could be helpful in understanding how mutations can affect the protein folding and BLM protein localization in nucleus, and its functions.
References
1) S. Akiko et al. (2010). Solution structure of the HRDC domain of human Bloom syndrome protein BLM. The Journal of Biochemistry. Volume 148; Issue 4.
2) Guo et al. (2007).Structural and functional analyses of disease-causing missense mutations in Bloom syndrome protein. Available from: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2094094/?tool=pubmed
3) Structure and function of the regulatory HRDC domain from human Bloom syndrome protein
2) Guo et al. (2007).Structural and functional analyses of disease-causing missense mutations in Bloom syndrome protein. Available from: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2094094/?tool=pubmed
3) Structure and function of the regulatory HRDC domain from human Bloom syndrome protein