This web page was produced as an assignment for Genetics 677, an undergraduate course at UW‐Madison.
Microarrays

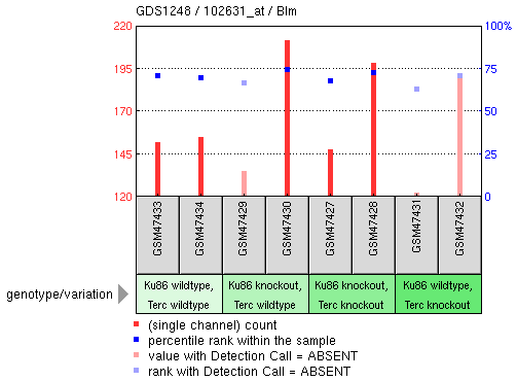
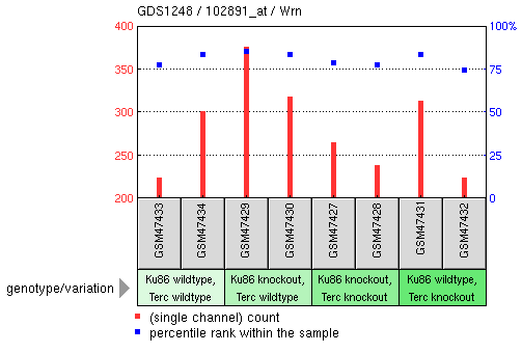
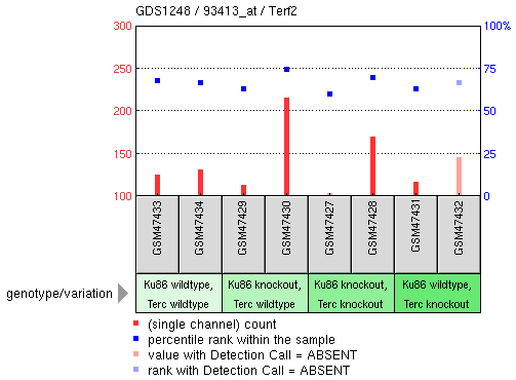
Microarray is an useful technique to compare gene expression levels under different conditions. Based on the information I obtained from BLM protein-protein interaction, I know that BLM interacts with TERF2, telomeric DNA-binding protein; therefore, I am interested in knowing the telomeric phenotype of BSyn patients. Entering "BLM telomere" under gene profiles query, I was able to get 2 results. I decided that one result that was related to Sgs1 gene in yeast irrelevant because it is not one of the homologues of human BLM. Below is one experiment showing gene expression levels in mouse with Terc and/or Ku86 knockout. The authors tested many genes, but instead of showing each gene's expression levels, I decided to look at two particular genes (BLM and WRN) and their expression profiles under each condition.
Experiment: Expression profiling of mus musculus (mouse) male germ cells lacking the telomerase RNA component, Terc, or the DNA repair/telomere binding protein, Ku86, or both.
Analysis
Even though BLM gene was shown to express in numerous experiments, I was particularly interested in studying the interaction between BLM protein, TERF2 and WRN protein (Werner Syndrome ATP-dependent protein). Both WRN and BLM have helicase activity and are found co-localized and interacted with TERF2 in vivo [2]. A number of studies has shown that Alternative Lengthening Telomere (ALT) is used by temolerase-negative, immortalized eukaryote cells to elongate or maintain the length of telomere [3].
Interestingly, in the last set of the test (Ku86 wildtype, Terc knockout), sample GSM47431 (from mus musculus) showed low or no expression of Blm while the expression of Terf2 was relatively higher than that of Blm. Meanwhile, Wrn was obviously expressed and had much higher expression than that of Blm and Terf2 when Terc was nonfunctional. As Terc was being knockout, there was presumably no telomerase activity in the mouse cells. Although it was not clear whether or not ALT was triggered in the mouse cells when Terc was being knockout, it is possible that Wrn was upregulated to take part in a mechanism maintaining the stability or length of mouse chromosomal telomere. Notably, it is impossible to summarize the functions Blm or Wrn in cells in absence of telomerase; therefore, more experiments need to be conducted in order to better understand Blm and Wrn roles in maintaining telomere stability.
Interestingly, in the last set of the test (Ku86 wildtype, Terc knockout), sample GSM47431 (from mus musculus) showed low or no expression of Blm while the expression of Terf2 was relatively higher than that of Blm. Meanwhile, Wrn was obviously expressed and had much higher expression than that of Blm and Terf2 when Terc was nonfunctional. As Terc was being knockout, there was presumably no telomerase activity in the mouse cells. Although it was not clear whether or not ALT was triggered in the mouse cells when Terc was being knockout, it is possible that Wrn was upregulated to take part in a mechanism maintaining the stability or length of mouse chromosomal telomere. Notably, it is impossible to summarize the functions Blm or Wrn in cells in absence of telomerase; therefore, more experiments need to be conducted in order to better understand Blm and Wrn roles in maintaining telomere stability.
References
1) Gene Expression Omnibus
2) Opresko, P.L. et al. (2002). Telomere-binding protein TRF2 binds to and stimulates the Werner and Bloom Syndrome helicases. The Journal of Biological Chemistry. Accessed April 16, 2011. Available from http://www.jbc.org/content/277/43/41110.long#ref-list-1
3) Bhattacharyya, S. et al. (2010). Unwinding protein complexes in Alternative Telomere Maintenance. J Cell Biochem. 109(1): 7–15. doi:10.1002/jcb.22388.
2) Opresko, P.L. et al. (2002). Telomere-binding protein TRF2 binds to and stimulates the Werner and Bloom Syndrome helicases. The Journal of Biological Chemistry. Accessed April 16, 2011. Available from http://www.jbc.org/content/277/43/41110.long#ref-list-1
3) Bhattacharyya, S. et al. (2010). Unwinding protein complexes in Alternative Telomere Maintenance. J Cell Biochem. 109(1): 7–15. doi:10.1002/jcb.22388.