This web page was produced as an assignment for Genetics 677, an undergraduate course at UW‐Madison.
About Bloom's Syndrome and BLM
Bloom's Syndrome
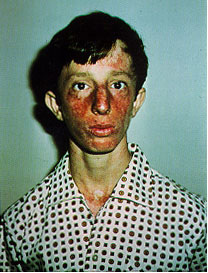
Bloom's Syndrome (BSyn) is a rare autosomal
recessive genetic disorder that is characterized by growth retardation that
leads to short stature, sun‐sensitive skin changes limited to face and dorsa of hands and forearms, paucity of
subcutaneous fat tissues throughout infancy and early childhood, and immunodeficiency
that are associated with otitis media and pneumonia [1], [2], [4]. However, the
most striking characteristic of BSyn is the increased risk of cancer
development. People with BSyn are likely to develop any cancers that can be
found in the general population, but at an early age, and they often develop
more than one type of cancer in their lives [1]. Since BSyn is inherited in an
autosomal recessive manner, both male and female are equally likely to get this disease.
Women with Bsyn might be fertile but they experience menopause prematurely; while men with BSyn are usually infertile and cannot
produce sperm [1].
Prevalence of Bloom's Syndrome
Due to fact that BSyn is a very rare genetic disorder, only a few cases of Bloom's Syndrome have been reported since it was first described by Dr. David Bloom in 1954 [2]. However, Bloom's Syndrome is considered relatively less rare among people with Ashkenazi Jewish background in Central and Eastern European [1]. Among the 265 individuals diagnosed with BSyn, 72 of them are of Ashkenazi Jewish background [2]. One particular mutation, 6-bp deletion and 7-bp insertion, is found to be prevalent among the Ashkenazi Jews and this particular mutation introduces stop codon within the open reading frame of BLM gene, thus resulting in premature translation termination and production of truncated protein [2]. Also, this mutant allele has an approximate frequency of 31% in individuals with BSyn and one or both their parents are Ashkenazi Jews [3], [4], (German, 2001).
BLM Gene
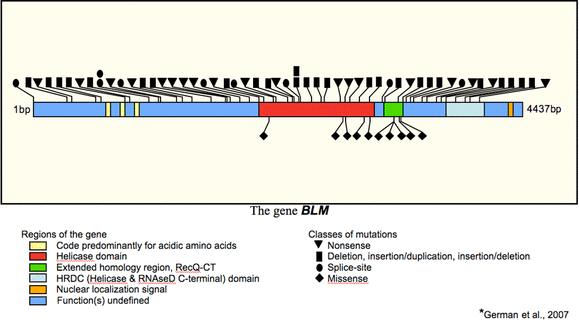
Figure 3: Gene BLM on chromosome 15
Locating on chromosome 15, in band q26.1, BLM
gene directs the production of a protein with a helicase domain that is 40%-45% of that in RecQ
helicase family [2]. RecQ helicase is an enzyme that binds to and unwinds double
helix DNA during DNA replication and DNA repairs [1].
So far, Bloom's Syndrome is known to be caused by mutations in only one specific gene- the BLM gene; however, as many as 60 mutations in BLM gene have been recognized [2], (German, 2001). Figure 3 on the right shows types of mutations that have been identified in different regions of BLM gene [4]. Oftentimes, mutations such as insertions and deletions cause frameshift and elimination of BLM protein's nuclear localization signals that is encoded by the 3' end of the gene, and hence resulting in the absence of BLM protein in nucleus [2].
Since normal BLM protein interacts with other proteins to maintain genome integrity and represses the occurrence of sister chromatids exchange, mutations in BLM gene cause genome instability: high occurrence of chromosome breaks, symmetric quadriradial chromatid interchange between homologous chromosomes and sister chromatid exchanges [1], [2]. BSyn can be clinically diagnosed via cytogenetic analysis, in which any cell types that can be cultured in vitro can be analyzed to confirm or rule out suspicion of BSyn. Since an unusually high sister chromatid exchange rate is the hallmark of BSyn, clinical diagnosis for BSyn often utilizes sister chromatid exchange (SCE) analysis to determine the frequency of SCEs in one's cultured cells [2]. An example of blood lymphocytes from an individual with BSyn was shown below (figure on the left). The BSyn blood lymphocytes show elevated SCE rate, and in fact, SCEs happen 10 times more frequently in BSyn cells than in normal somatic cells (Amor‐Gueret, 2006). Because of the hyperrecombinability and hypermutability, people with BSyn are predisposed to spontaneous mutations that later lead to cancer development (German, 2001).
So far, Bloom's Syndrome is known to be caused by mutations in only one specific gene- the BLM gene; however, as many as 60 mutations in BLM gene have been recognized [2], (German, 2001). Figure 3 on the right shows types of mutations that have been identified in different regions of BLM gene [4]. Oftentimes, mutations such as insertions and deletions cause frameshift and elimination of BLM protein's nuclear localization signals that is encoded by the 3' end of the gene, and hence resulting in the absence of BLM protein in nucleus [2].
Since normal BLM protein interacts with other proteins to maintain genome integrity and represses the occurrence of sister chromatids exchange, mutations in BLM gene cause genome instability: high occurrence of chromosome breaks, symmetric quadriradial chromatid interchange between homologous chromosomes and sister chromatid exchanges [1], [2]. BSyn can be clinically diagnosed via cytogenetic analysis, in which any cell types that can be cultured in vitro can be analyzed to confirm or rule out suspicion of BSyn. Since an unusually high sister chromatid exchange rate is the hallmark of BSyn, clinical diagnosis for BSyn often utilizes sister chromatid exchange (SCE) analysis to determine the frequency of SCEs in one's cultured cells [2]. An example of blood lymphocytes from an individual with BSyn was shown below (figure on the left). The BSyn blood lymphocytes show elevated SCE rate, and in fact, SCEs happen 10 times more frequently in BSyn cells than in normal somatic cells (Amor‐Gueret, 2006). Because of the hyperrecombinability and hypermutability, people with BSyn are predisposed to spontaneous mutations that later lead to cancer development (German, 2001).
References:
1) Genetics Home Reference: Your guide to understanding genetic conditions. Retrieved January 29, 2011from http://ghr.nlm.nih.gov/condition/bloom‐syndrome
2) Sanz, M.M., German, J. (2010). Bloom's syndrome. GeneReviews [Internet]. Retrieved January 25, 2011 from http://www.ncbi.nlm.nih.gov/books/NBK1398/
3) German, J., Ellis, N. A. (2005). Bloom syndrome. The McGraw‐Hill. Retrieved February 2, 2011 from http://books.mcgraw‐hill.com/getommbid.php?isbn=0071459960&template=ommbid&c=30
4) Weill Cornell Medical centert. Bloom's Syndrome Registry. Retrieved January 25, 2011 from http://weill.cornell.edu/bsr/
5) German, J. Bloom’s syndrome. Encyclopedia of Genetics. 229–230, 2001
6) Amor‐Gueret, M. Bloom syndrome, genomic instability and cancer: the SOS‐like hypothesis. Cancer Letters. 236:1–12, 2006.
7) Banner Photo: http://www.sciencephoto.com/images/download_lo_res.html?id=670030948
8) Figure 1: http://web.mit.edu/biology/guarente/human/human.html
9) Figure 2: http://weill.cornell.edu/bsr/
10) Figure 3: http://weill.cornell.edu/bsr/genetics/
11) Figure 4 and 5: http://weill.cornell.edu/bsr/lab_diagnosis/
2) Sanz, M.M., German, J. (2010). Bloom's syndrome. GeneReviews [Internet]. Retrieved January 25, 2011 from http://www.ncbi.nlm.nih.gov/books/NBK1398/
3) German, J., Ellis, N. A. (2005). Bloom syndrome. The McGraw‐Hill. Retrieved February 2, 2011 from http://books.mcgraw‐hill.com/getommbid.php?isbn=0071459960&template=ommbid&c=30
4) Weill Cornell Medical centert. Bloom's Syndrome Registry. Retrieved January 25, 2011 from http://weill.cornell.edu/bsr/
5) German, J. Bloom’s syndrome. Encyclopedia of Genetics. 229–230, 2001
6) Amor‐Gueret, M. Bloom syndrome, genomic instability and cancer: the SOS‐like hypothesis. Cancer Letters. 236:1–12, 2006.
7) Banner Photo: http://www.sciencephoto.com/images/download_lo_res.html?id=670030948
8) Figure 1: http://web.mit.edu/biology/guarente/human/human.html
9) Figure 2: http://weill.cornell.edu/bsr/
10) Figure 3: http://weill.cornell.edu/bsr/genetics/
11) Figure 4 and 5: http://weill.cornell.edu/bsr/lab_diagnosis/